Latest California Healthline Stories
KFF Health News' 'What the Health?': Kennedy Cancels Vaccine Funding
The Health and Human Services secretary is winding down nearly $500 million in mRNA research funding, citing false claims that the technology is ineffective against respiratory illnesses — and notching a victory for critics of the covid vaccines. And President Donald Trump is demanding drugmakers drop their prices, quickly, but it’s unclear how he could make them comply. Lauren Weber of The Washington Post, Sandhya Raman of CQ Roll Call, and Sarah Karlin-Smith of the Pink Sheet join KFF Health News’ Emmarie Huetteman to discuss these stories and more.
Sorting Out Covid Vaccine Confusion: New and Conflicting Federal Policies Raise Questions
Should you get vaccinated? Will your insurer pay for it? And will you still be able to find a vaccine? KFF Health News tries to sort out where things stand.
Aclarando la confusión sobre las vacunas contra covid-19
Las agencias dentro del Departamento de Salud y Servicios Sociales responsables de especificar quién debe vacunarse no están necesariamente sincronizadas, emitiendo recomendaciones en apariencia contradictorias.
KFF Health News' 'What the Health?': Next on Kennedy’s List? Preventive Care and Vaccine Harm
Robert F. Kennedy Jr., the secretary of Health and Human Services, is eyeing an overhaul of two more key entities as part of his ongoing effort to reshape health policy. And President Donald Trump signed an executive order last week that would enable localities to force some homeless people into residential treatment. Anna Edney of Bloomberg News, Joanne Kenen of the Johns Hopkins Bloomberg School of Public Health and Politico Magazine, and Shefali Luthra of The 19th join KFF Health News’ Julie Rovner to discuss these stories and more. Also, Rovner interviews Sara Rosenbaum, one of the nation’s leading experts on Medicaid, to mark Medicaid’s 60th anniversary this week.
KFF Health News' 'What the Health?': Here Come the ACA Premium Hikes
Medicaid may have monopolized Washington’s attention lately, but big changes are coming to the Affordable Care Act as well. Meanwhile, Americans are learning more about what’s in Trump’s big budget law, and polls suggest many don’t like what they see. Julie Appleby of KFF Health News, Jessie Hellmann of CQ Roll Call, and Alice Miranda Ollstein of Politico join KFF Health News’ Julie Rovner to discuss these stories and more. Also this week, Rovner interviews historian Jonathan Oberlander to mark Medicare’s 60th anniversary.
KFF Health News' 'What the Health?': The Senate Saves PEPFAR Funding — For Now
The Senate narrowly approved the Trump administration’s request to claw back about $9 billion for foreign aid and public broadcasting but refused to cut funding for the international AIDS/HIV program PEPFAR. Meanwhile, a federal appeals court ruled that West Virginia can ban the abortion pill mifepristone, which could allow states to block other FDA-approved drugs. Joanne Kenen of the Johns Hopkins Bloomberg School of Public Health and Politico Magazine, Shefali Luthra of The 19th, and Sandhya Raman of CQ Roll Call join KFF Health News’ Julie Rovner to discuss these stories and more.
What RFK Jr. Isn’t Talking About: How To Make Vaccines Safer
Vaccines are under fire from the top of the Trump administration. Federal programs to monitor them and make them safer have always been underfunded.
A Revolutionary Drug for Extreme Hunger Offers Clues to Obesity’s Complexity
A new drug is helping families who’ve spent years padlocking fridges, chaining garbage cans, and hiding food as their children with Prader-Willi syndrome deal with unrelenting hunger. But additional progress — and a broader understanding of obesity — is now under threat as the government dismantles the pipeline for promising new research.
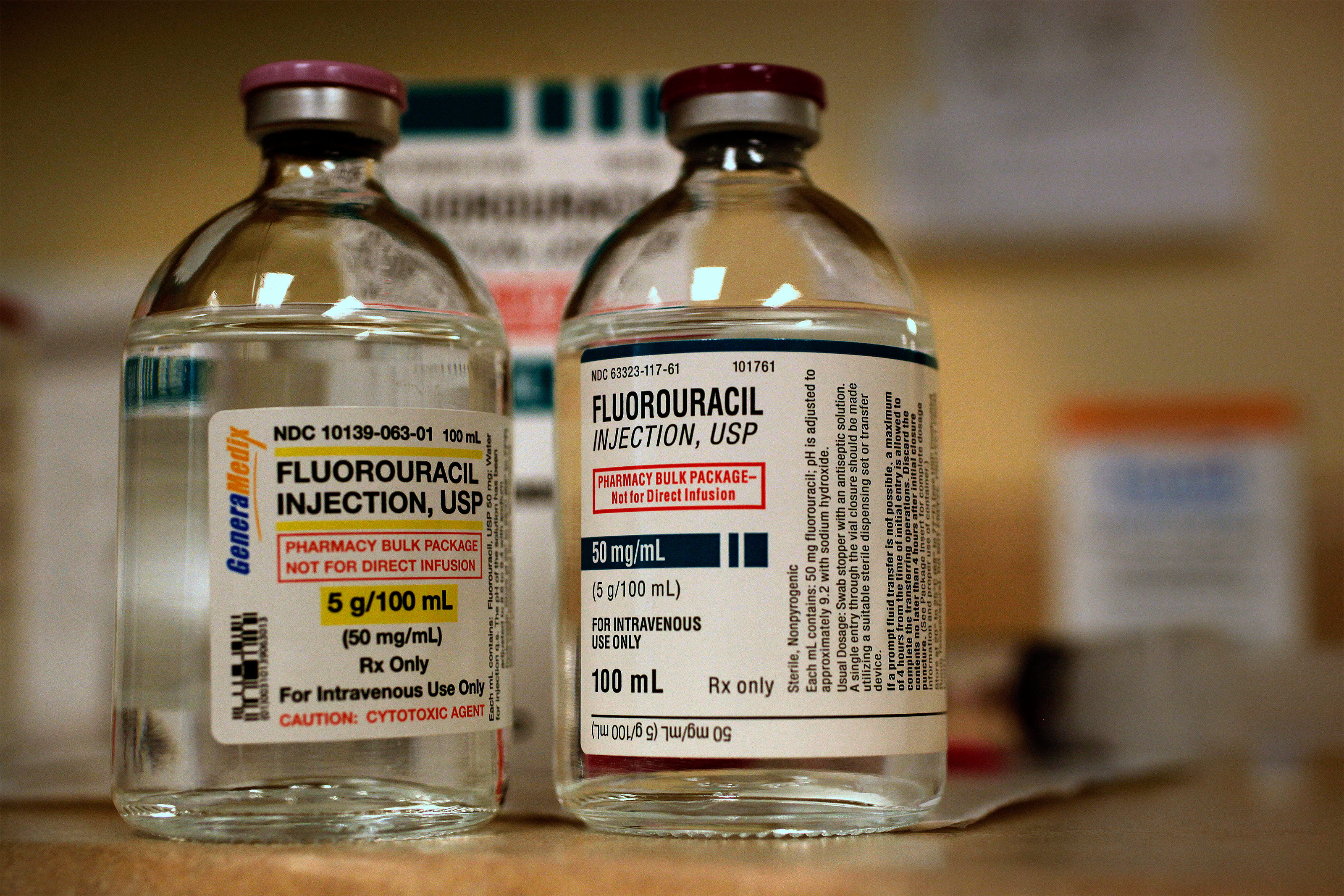
Two Patients Faced Chemo. The One Who Survived Demanded a Test To See if It Was Safe.
Worried that President Donald Trump’s FDA might not act, a panel of cancer experts recommended that doctors consider testing before dosing patients with a commonly used but sometimes deadly cancer drug. It came too late for many patients.
Silence on E. Coli Outbreak Highlights How Trump Team’s Changes Undermine Food Safety
Food safety inspections are being scaled back and the public was not notified after an investigation into E. coli contamination.